Researchers from the Autophagy Laboratory of the Margarita Salas Center for Biological Research have identified the molecular mechanism responsible for blocking autophagy in age-related macular degeneration in a paper published in the journal Molecular Neurodegeneration. The research also demonstrates the neuroprotective role of urolithin A in the murine model of this disease.
Downregulation of autophagy, the main intracellular recycling system, is one of the key hallmarks of aging. Its malfunction has been associated with diseases such as age-related macular degeneration (AMD). This pathology affects around 200 million people worldwide, and it is expected to double by 2040 due to population aging. AMD is characterized by a progressive degeneration of the macula, located in the central region of the retina, and is responsible for color and high-resolution vision, which can culminate in loss of central vision.
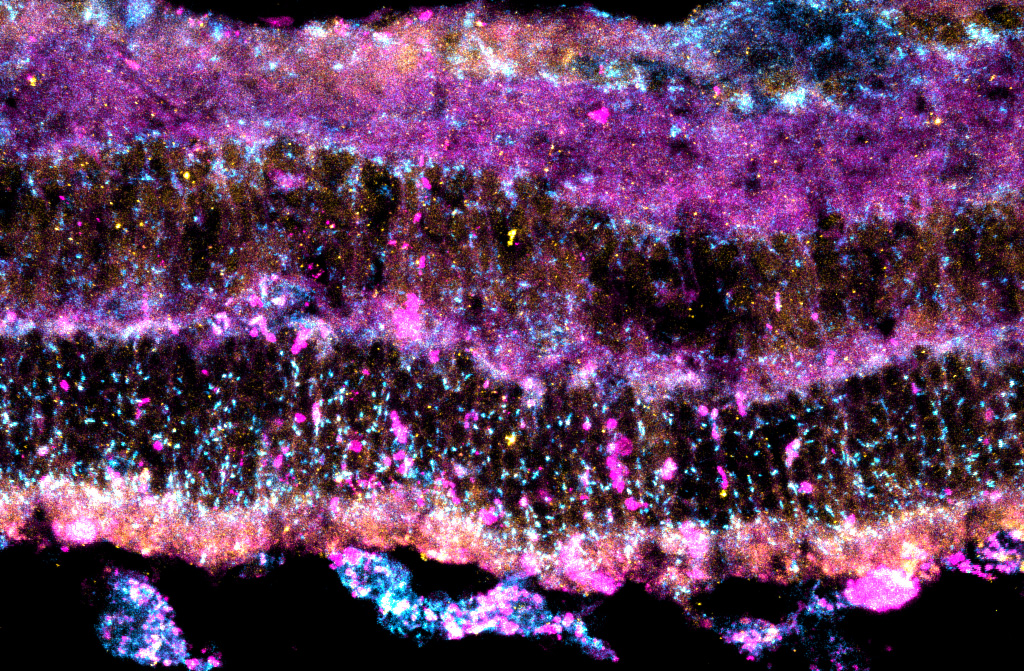
Jiménez-Loygorri et al. have demonstrated, using a pharmacological model of AMD in mice, that the retinal pigment epithelium (RPE; the main cell type affected in AMD) suffers a severe blockage in the degradation of damaged intracellular components via autophagy. This alteration leads to the accumulation of protein aggregates, oxidized lipid membrane, and dysfunctional mitochondria, causing an intracellular collapse that triggers the death of the RPE. The RPE provides nutritional support to all other retinal neurons, including photoreceptors, and this ultimately leads to vision loss, which is also observed in the mouse model.
The study, which combined functional and molecular assays, concluded that this autophagy blockage is due to permeabilization (formation of small pores) in the membrane of lysosomes, the organelle responsible for degrading and recycling intracellular waste.
In the AMD model, the researchers also studied the neuroprotective potential of urolithin A (UA), a natural compound derived from pomegranate metabolites. Previous studies from the Autophagy lab have shown that UA can reduce age-associated inflammation through its ability to induce mitophagy, the selective recycling of damaged or superfluous mitochondria via autophagy. Although UA can also restore and further induce mitophagy in the AMD model, genetic manipulations showed that it was dispensable for its neuroprotective effect.
The now-published work shows that UA can also promote the recycling of damaged lysosomes through lysophagy, thus restoring autophagic flux and cellular homeostasis. This process is mediated by the protein SQSTM1/p62, which can recognize damaged lysosomes, tag them, and signal that they should be recycled. Treatment with UA therefore promotes RPE survival, and proper photoreceptor function and prevents vision loss in the murine model of AMD.
This discovery opens the door to the use of UA for the treatment of age-related diseases characterized by lysosomal damage, such as AMD or Alzheimer's disease.
Reference: Urolithin A promotes p62-dependent lysophagy to prevent acute retinal neurodegeneration. Juan Ignacio Jiménez-Loygorri#, Álvaro Viedma-Poyatos, Raquel Gómez-Sintes, Patricia Boya# (2024) Molecular Neurodegeneration. DOI: https://doi.org/10.1186/s13024-024-00739-3

