The One Earth Series
Alicia Bravo and Manuel Espinosa. Centro de Investigaciones Biológicas Margarita Salas, Consejo Superior de Investigaciones Científicas (CSIC). Ramiro de Maeztu, 9. 28040 Madrid, Spain.
This series comprises four Chapters devoted to describing the One Earth concept. We shall discuss the relationships between two separate, albeit intimate, beings sharing the same niches in many cases: Humans and Bacteria. Why are we afraid of Bacteria? Will Bacteria kill us all? How can we defend ourselves from their deadly attacks? Must we share the same niches? These and many other questions will be raised and discussed here. But we can anticipate the answer: We must share and co-exist. Else, Humans will be doomed.
Introduction
In Chapter II we discussed the relevance of being cautious when we get symptoms of an infectious disease and keeping a distance from our relatives to avoid being contagious to them. We discussed that the misuse of antibiotics in humans, cattle, fisheries, and crops has led to the selection of bacterial species resistant to multiple antibiotics, the superbugs. We have scarce or no treatment when such highly resistant bacteria infect us. In many cases, the treatment may be very expensive and lengthy. It is important to remember that we are dealing with highly resistant pathogenic bacteria. In other cases, the infections are caused by opportunistic bacteria, that is bacteria that are usually harmless but can turn into pathogenic when our immune system is weak. The superbugs have been selected by the improper use (and abuse in many cases) of antibiotics, and now we ask how we arrived at the present situation. Antibiotic resistance is especially dramatic in hospital-acquired (nosocomial) infections because any small surgery or anti-cancer treatment may be followed, in many cases, by severe bacterial infections, which will be treated with high dosages of potent antibiotics. This will create a loop that will lead, sooner or later, to the selection of bacteria also resistant to such antibiotics.
The Scenario
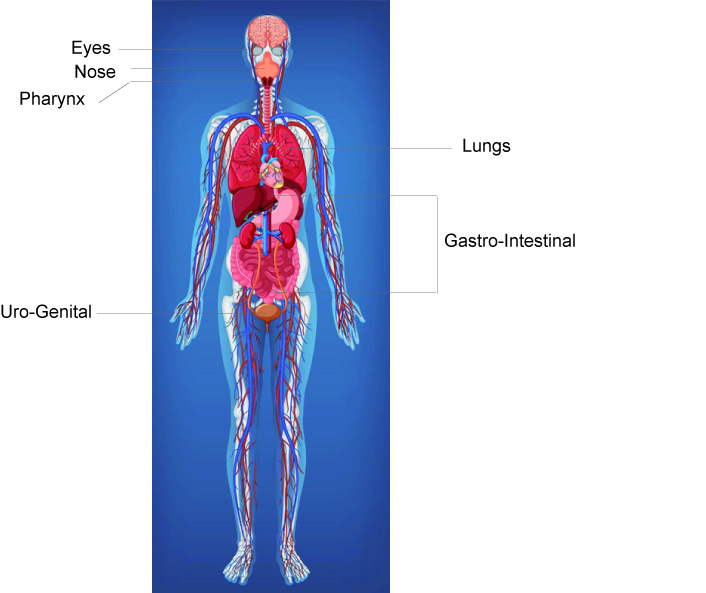
Let us contemplate the panorama we are surrounded by: the human mucosal surfaces, or just the mucosa (Figure). The mucosa is continuous with the skin at body openings such as the eyes, eyelids, ears, inside the nose (nares), inside the mouth, lips, the genital areas, the urethral opening, and the anus. The mucosal surfaces also include the digestive, the urogenital, and the respiratory tracts. Mucous membranes exist all over our body, lining our canals and many of our internal organs. It is designed to protect the organs and canals from external and internal irritants. The mucosa alone comprises around 400 square meters (a nice house, plus a small garden, right?). Bacteria colonize these surfaces. The most bacteria-populated areas of the mucosa are the nares, mouth, and gut. All the bacteria that live in the mucosa participate actively in our everyday activities and protect our organs from external aggressions when we are in healthy conditions. The supervising task of keeping a peaceful compromise between our colonizing bacteria and the epithelial cells of the mucosa is exerted by our immune system. Now, we can understand why the high-risk population includes people having immature (children below 5), defective (elders above 70), and weak or suppressed (e.g. cancer treatment therapies) immune systems.
The Present Vista
In nature, many antibiotics are produced by some bacterial species to compete with (and eventually, kill) other bacterial populations with whom they share the same niche. The antibiotics produced by the former bacteria do not reach the concentrations that are normally used in clinical treatments to kill the bacteria that are causing an infection. Further, the exposure of the competing bacteria to the naturally present soil antibiotics does not take as long as in the clinical treatments. At present, antibiotic resistance is inevitable and irreversible because it is the result of a brutal selection posed by us, humans, on the bacterial world over the years. These newly selected bacteria have been compelled to a fast and forced evolution during exposure to high doses of antibiotics. The consequences are the following: first, the present antibiotics are losing effectiveness; second, the bacterial infections will be more difficult and expensive to treat, and third, epidemics will be hardly controlled. The World Health Organization (WHO) predicted a human toll of ~10 million deaths per year globally by 2050 due to the superbugs (report of Nov, 17th, 2021 (https://www.who.int/news-room/fact-sheets/detail/antimicrobial-resistance). WHO also demanded an urgent response to the subject. However, countries' response has been, unsurprisingly, sloppy and insufficient. In 2022, it was published in one of the most influencing scientific journals that an analysis carried out in 204 countries estimated that up to five million people died in 2019 (before the official start of the COVID-19 pandemic) from illnesses associated with bacteria resistant to antibiotics. In the same year, AIDS and malaria caused 860,000 and 640,000 deaths, respectively (Murray et al., 2022). We are under a clear case of attaining the so-called “worst scenario”, which has been increased by the COVID-19 viral pandemic: most hospital treatments include the routine use of antibiotics (in addition to self-treatments in many countries) because of the risk of secondary bacterial pneumonia after viral infections, which has led to a further selection of superbugs. Moreover, a recent report made by the Antimicrobial Resistance Industry Alliance shows that there has been a serious “brain drainage” of professionals working on the subject as the result of both governments and pharmaceutical companies investing less in developing antimicrobial drugs (see “Further Readings”, below).
Finding the Equilibrium
The health crisis associated with the selection of superbugs should be conceived as a dilemma with evolutionary, ecological, and economic implications. Furthermore, we must take into account the self-believed “supremacy” of humans over other beings and, in consequence, “our right” to eliminate those competing organisms that are prejudicial to us in any sense, like fungi, ‘bad’ weeds, spiders, insects, mice, and rats, to name a few. The predatory nature of humans has led to the extinction of numerous species. This, together with the super-development of crops, cattle, poultry, and fisheries, has important consequences on the global ecology and biodiversity of our planet, on climate change, and, as a consequence, on the evolution of the biosphere. We have to learn that bacteria play a crucial role in these matters. A scientific warning has been issued on the importance of microorganisms in climate change
Then, our next question would be: is there any turning back to previous situations? Let’s advance a hopeful answer: maybe yes.
Further Readings:
In Spanish:
Alós, J.I. (2015) Resistencia bacteriana a los antibióticos: una crisis global. Enferm. Infecc. Microbiol. Clin. 33: 692-699.
In English:
Bravo A, Moreno-Blanco A & Espinosa M (2023) One Earth: The Equilibrium between the Human and the Bacterial Worlds. Int. J. Mol. Sci. 24: 15047.
Bravo A, Ruiz-Cruz S, Alkorta I & Espinosa M (2018) When Humans Met Superbugs: Strategies to Tackle Bacterial Resistance to Antibiotics. BioMol. Concepts 9: 216-226.
Cavicchioli R, Ripple WJ, Timmis KN, et al. (2019) Scientists’ warning to humanity: Microorganisms and Climate Change. Nat. Rev. Microbiol. 17: 569-586.
Murray CJL, Ikuta KS, Sharara F, et al. (2022) Global Burden of Bacterial Antimicrobial Resistance in 2019: A Systematic Analysis. Lancet 399: 629-655.
https://wellcome.org/news/climate-change-antimicrobial-resistance. Will climate change lead to more drug-resistant infections?
Leaving the Lab: https://www.amrindustryalliance.org/newsroom/leaving-the-lab-tracking-the-decline-in-amr-rd-professionals/
Acknowledgements
This series attempts to communicate our research at the Centro de Investigaciones Biológicas Margarita Salas, CSIC and is part of the Grant I+D+i PID2019-104553RB-C21, funded by MICIU/AEI/10.13039/501100011033. Thanks are due to María del Carmen Fernández and Mónica Fontenla for their help during the elaboration of this series.
Other chapters:
Chapter I: link.
Chapter II: link.
Chapter IV: link.

