Group Leader/s
intro
Dr. Maria Montoya (MM) has worked in pig immunology for the last thirteen years, studying immune responses to viruses affecting pigs, such influenza virus, and generating novel vaccine strategies. She has a long track record in viral immunology in different species and systems with international collaborations worldwide. MM has worked with a variety of pathogens under SAPO4 conditions for 15 years. She has been PI on several successful grants and published more than 60 papers in peer reviewed journals (Nature, Journal of Immunology, Blood, Veterinary Research, Plos Pathogens, Scientific Reports, Journal of Virology, Plos One...). MM support the efforts to improve the role of women in science as well as participate in outreaching activities for the general public.
Aims and objectives of the group
In the last decades, the mouse model tremendously contributed to the progress of research in immunology. However, it is worth noticing that research in large/unconventional animals has given pivotal information to immunology in general. The need for translational immunological research, the development of new animal models more adapted to the scientific question, and the acquisition of new knowledge in comparative immunology is increasing in importance every year.
The main aim of the group is to provide new insights into immunological host-pathogen interactions by studying the immune system of the pigs in the context of natural relevant viral infections. All previous experience has been used to study inflammatory mechanisms of SARS-CoV-2. Pigs will be used as model for cellular therapies or viral infections.
Viral infections continue to cause considerable morbidity and mortality around the world. Besides its importance as a zoonotic agent, Swine Influenza Virus (SwIV) is a relevant porcine respiratory disease. African swine fever virus (ASFV) causes severe disease in domestic pigs resulting in up to 100% mortality and there is no vaccine. SARS-CoV-2 is a new type of coronavirus causing the actual pandemic worldwide. New approaches are required to fight viral diseases, which can only be informed by a deeper knowledge of immune responses during host-pathogen interactions. The work in the group is directed into a better understanding of the immune responses required for protection against viral diseases.
Our team have applied reverse vaccinology to identify SLA-I and SLA-II T-cell epitopes from SwIV H1N1. Recognition of overlapping peptides from H1N1 revealed a novel SLA class II restricted epitope in NP. This conserved novel epitope could be the base for further vaccine approaches against H1N1 SwIV in pigs (Fig 1).

Fig 1
Extracellular vesicles (EVs) have proven to be a promising new vaccination platform in situations in which conventional approaches have not been completely successful. Serum EVs from infected pigs using two different ASFV viruses were characterized. Proteomic analysis revealed few specific proteins from ASFV in the EVs, but 942 swine proteins were detected in all EV preparations. Most of these were related to coagulation cascades. The results obtained contribute to a better understanding of ASFV pathogenesis and immune/protective responses in the host.
The pathophysiology of SARS-CoV-2 has not been well characterized. Particularly, the reason why some people develop an exacerbated inflammation leading to disease severity remains obscure. We are working to build up a cellular platform designed to establish viral determinants that may modulate inflammation and/or cell death (Fig 2). These findings will be used to develop novel approaches to counteract inflammation as a possible treatment.
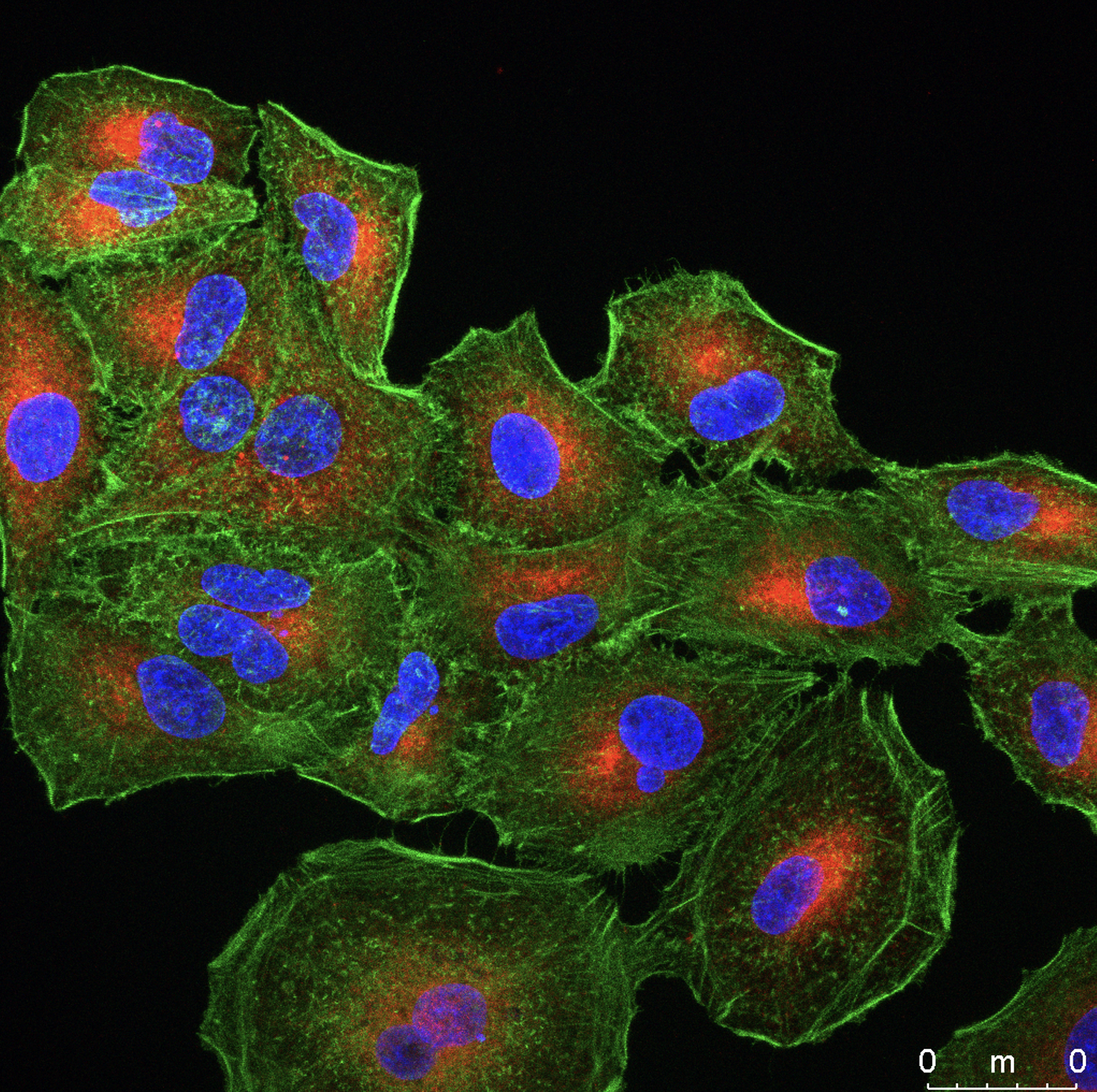
Fig 2
Members
| María Montoya González |
| Sergio Barata Garcia |
| Blanca Dies Lopez-Ayllon |
| Larysa Muzykina |
| Sofia Perez Ramos |
| Laura Mendoza Garcia |
| Ana de Lucas Rius |
| Alicia Carmen Marin Gomez |
| Juozas Grigas |

Selected Publications
P. J. Alcolea, J. Larraga, D. Rodríguez-Martín, A. Alonso, F. J. Loayza, J. M. Rojas, S. Ruiz-García, A, Louloudes-Lázaro, A. B. Carlón, P. J. Sánchez-Cordón, P. Nogales-Altozano, N. Redondo, M. Manzano, D. Lozano, Jesús Palomero, M. Montoya, M. Vallet-Regí, V. Martín, N. Sevilla and Vicente Larraga [2022]. Non-replicative antibiotic resistance-free DNA vaccine encoding S and N proteins induces full protection in mice against SARS-CoV-2
A. Sanluis-Verdes , P. Colomer-Vidal, F. Rodriguez-Ventura, M, Bello-Villarino, M. Spinola-Amilibia, E. Ruiz-Lopez, R. Illanes-Vicioso, P. Castroviejo, R. Aiese Cigliano, M. Montoya, P. Falabella, C. Pesquera, L. Gonzalez-Legarreta, E. Arias-Palomo, M. Solà, T. Torroba, C. F. Arias, F. Bertocchini [2022]. Wax worm saliva and the enzymes therein are the key to polyethylene degradation by Galleria mellonella
Natalia Redondo, Sara Zaldívar-López, Juan J Garrido, Maria Montoya [2021]. SARS-CoV-2 Accessory Proteins in Viral Pathogenesis: Knowns and Unknowns
Funding
Selected competitive grants from 2019:
1. “Cooperación en inmunología avanzada: capacitación de técnicas y diseño de curso de grado y postgrado”. Proyectos CSIC de cooperación I+COOP+2019 (COOPB20438) IP: M Montoya. 2019-2021. 23.550€

2. “Use of extracellular vesicles as immunogens for African Swine Fever Virus”. Proyecto de colaboración entre países de la UE. VetBioNet. IP: M Montoya. 2020. 63.550€

3. "Inflammation viral determinants in the cytokine storm within COVID-19". Proyecto financiado por la PTI Salud Global CSIC en la pandemia COVID-19 (COVID-19-117) IP: M Montoya. 2020. 117.000€

4. "Inflammation viral determinants in the cytokine storm within COVID-19". Proyecto financiado por la Junta de Andalucía en la pandemia COVID-19 (CV20-20089). IP: JJ Garrido. 2020. 80.000€

5. "Inflammation viral determinants in the cytokine storm within COVID-19". Proyecto financiado por la PTI Salud Global CSIC en la pandemia COVID-19 (COVID-19-117) IP: M Montoya. 2020. 439.150€.

6. ALBA synchrotron granted beamtime in 2022: MISTRAL (2022025607, proposer, 15 shifts). IP1: MA Oliva, IP2: M Montoya.

7. "Cytoskeleton and inflammation in viral infections: new options on the search of wide range anti-virals". Proyecto financiado por la Agencia Española de Investigación (Plan nacional) (PID2021-123399OB-I00). IP1: M Montoya, IP2: M Oliva. 2022. 278.300€.




8. "Improving biosecurity and welfare of swine farms and products by integration of non-invasive laboratory diagnostics, sensorics of the environment and big data" Proyecto financiado por la Agencia Española de Investigación para el fomento de la colaboración publico privada (CPP2021-008618) MCIN/AEI/ 10.13039/501100011033 y por la Unión Europea NextGenerationEU/PRTR. Coordinator: Antonio González de Bulnes López (1.287.150,50€). IP WP3: M Montoya. 2022. 235.734,50€.




9."Emerging porcine influenza and coronaviruses (EPICVIR)". Proyecto PCI2023-143414, financiado por MCIN/AEI/10.13039/501100011033 y por la Unión Europea Era-Net ICRAD (EU). Coordinadora: Dr. K. Van Reeth (1.744.00€). P3: M Montoya. 2023. 174.000€


10. “European Fund for Displaced Scientists Programme, Grant Forwarding and Cooperation Agreement" Proyecto financiado por ALLEA (EFDS-FL1-34) para científicos ukranianos desplazados por la guerra. IP: M Montoya. 2023. 24.960€

11. “Cooperation in Transcriptomics and Diagnostic. Design of postgraduate studies”. Proyectos CSIC de cooperación I+COOP+2022 (COOPB22065) IP: M Montoya. 2023-24. 40.000€

12. ALBA synchrotron granted beamtime in 2023: MISTRAL (proposer, 15 shifts). IP1: MA Oliva, IP2: M Montoya.

More info
MARIA MONTOYA GONZALEZ
Master in Immunology 1991-1992. University of Birmingham, UK.
PhD: 1997, Universidad Autonoma de Madrid, Spain
Post-doctoral and Fellow Scientist: 1998-2005. The Edward Jenner Institute. UK
Group leader: 2005-2014. Centre de Recerca en Sanitat Animal (CReSA). Barcelona, Spain
Group leader: 2014-2017. The Pirbright Institute. UK
Patents
EU patent “Exosomes and their use as vaccine”. P3249PC00. 2016. H. A. del Portillo, F. E. Borras, L.J. Fraile, S.R. Montaner, M. Montoya.
Patent subcontracted and in development by a company.
Spin-off company
Innovex Therapeutics SL. (https://innovexther.com/ )
Investigador Distinguido: 2018. Centro de investigaciones Biológicas (CIB), Madrid, Spain

